Description
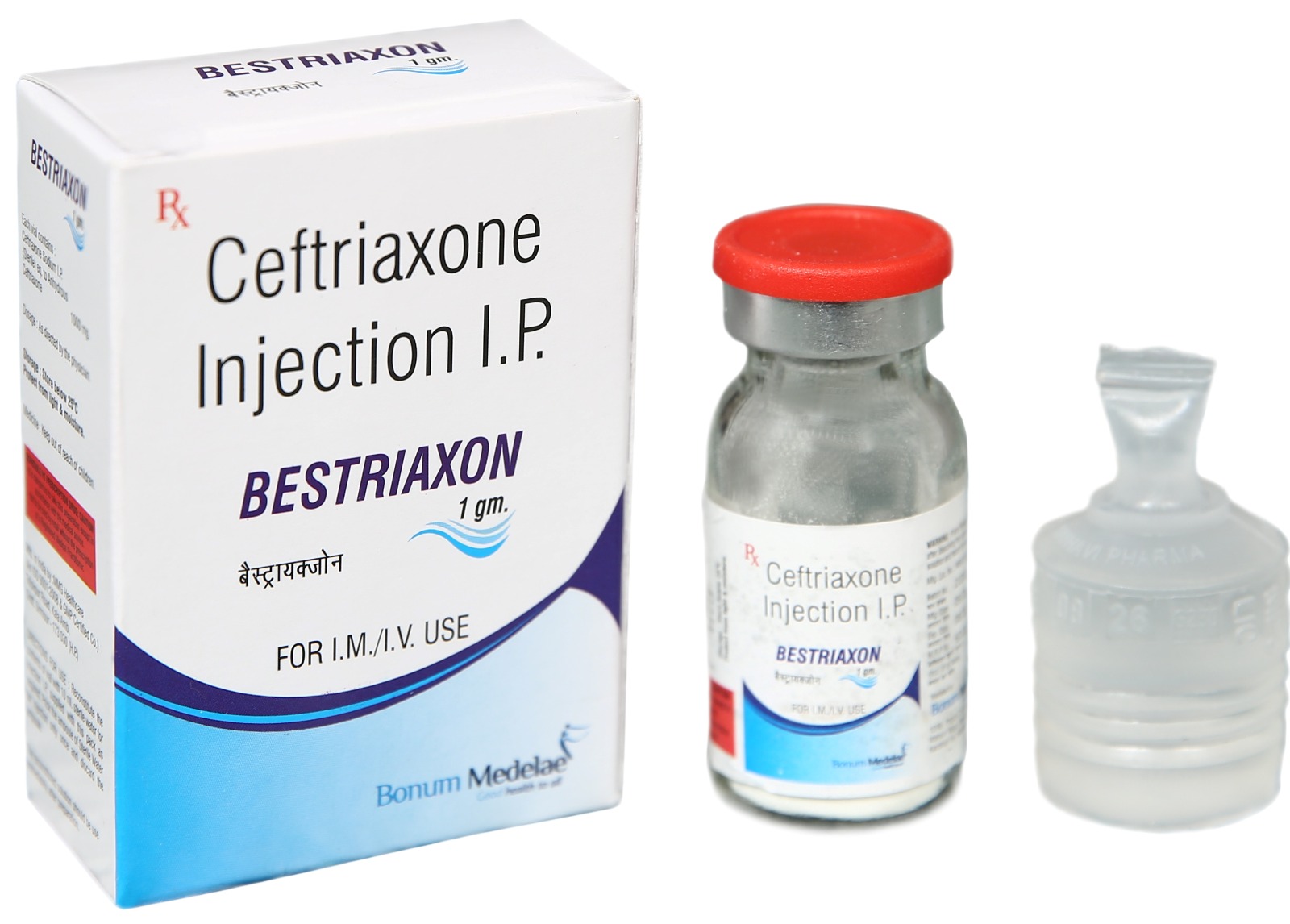
Bestriaxon Injection – Premium Antibiotic for Serious Bacterial Infections
Composition:
Bestriaxon Injection contains Ceftriaxone Sodium equivalent to Ceftriaxone 1g per vial (also available in 250mg, 500mg, and 2g strengths). Ceftriaxone is a third-generation cephalosporin antibiotic with broad-spectrum bactericidal activity against gram-positive and gram-negative bacteria.
Uses & Indications:
Bestriaxon Injection is indicated for the treatment of serious bacterial infections including:
• Respiratory tract infections (pneumonia, bronchitis)
• Urinary tract infections (UTIs)
• Skin and soft tissue infections
• Bone and joint infections (osteomyelitis, septic arthritis)
• Intra-abdominal infections (peritonitis)
• Meningitis and other central nervous system infections
• Septicemia (blood infections)
• Gonorrhea and pelvic inflammatory disease
• Surgical prophylaxis
• Bacterial infections in immunocompromised patients
Key Benefits:
✓ Broad-spectrum antibiotic – effective against multiple bacterial strains
✓ Powerful bactericidal action – kills bacteria rather than just inhibiting growth
✓ Long half-life – allows once or twice daily dosing
✓ High tissue penetration – reaches infection sites effectively
✓ Suitable for severe infections – hospital-grade antibiotic therapy
✓ Proven efficacy – trusted by healthcare professionals worldwide
✓ Works when oral antibiotics fail – injectable formulation ensures direct delivery
Administration & Dosage:
Bestriaxon Injection is administered via intramuscular (IM) or intravenous (IV) route under medical supervision. The dosage varies based on infection severity, patient age, weight, and kidney function:
• Adults: 1-2g once daily or in divided doses (maximum 4g daily)
• Children: 50-75 mg/kg body weight once daily or in divided doses
• Treatment duration: Typically 7-14 days depending on infection type and response
Always follow your physician’s prescribed dosage and complete the full course of treatment.
Precautions & Warnings:
⚠ Not for self-administration – must be given by qualified healthcare professionals
⚠ Inform your doctor about allergies to penicillins or cephalosporins
⚠ Use with caution in patients with kidney or liver disease
⚠ May interact with calcium-containing solutions
⚠ Not recommended during pregnancy and breastfeeding without medical consultation
⚠ Complete blood count monitoring may be required during prolonged therapy
⚠ May cause pseudomembranous colitis – seek immediate medical attention if severe diarrhea occurs
Possible Side Effects:
Common side effects may include:
• Injection site reactions (pain, redness, swelling)
• Diarrhea or loose stools
• Nausea and vomiting
• Skin rash or itching
• Elevated liver enzymes
• Changes in blood cell counts
Serious side effects (seek immediate medical help):
• Severe allergic reactions (anaphylaxis)
• Severe skin reactions (Stevens-Johnson syndrome)
• Severe diarrhea with blood (C. difficile infection)
• Jaundice or dark urine
• Unusual bleeding or bruising
Storage:
Store below 25°C in a cool, dry place. Protect from light. Reconstituted solution should be used immediately or stored as per manufacturer’s guidelines. Keep out of reach of children.
Why Choose Bestriaxon Injection?
Bestriaxon is manufactured under strict quality controls meeting international pharmaceutical standards. It provides reliable and effective treatment for serious bacterial infections when immediate and potent antibiotic therapy is required. Trust Bestriaxon for hospital-grade infection management.
Keywords: Bestriaxon Injection, Ceftriaxone injection, antibiotic injection, bacterial infection treatment, third-generation cephalosporin, broad-spectrum antibiotic, pneumonia treatment, UTI injection, septicemia treatment, meningitis antibiotic, IV antibiotic, hospital antibiotic, prescription antibiotic injection, Ceftriaxone sodium injection, infection treatment, injectable antibiotic

Reviews
There are no reviews yet.